Scinvivo secures 1 Million investment for in human clinical trials & CE
The urologists who reviewed the technology see this as a likely game-changer in their diagnostic routine with the potential to become the golden stand
Press release closing 1 Million investment Scinvivo
Eindhoven, March 10th, 2022
Financing secured towards in human clinical trials & CE
Scinvivo, a MedTech startup based in Eindhoven, has closed an investment round of 1 Million Euro from NextStage Early Stage Fund (NSESF), Innovatiefonds Noord-Holland, and current shareholders. With this investment, Scinvivo has secured the development of in human clinical trials and CE certification. Scinvivo is on a mission to make cancer diagnostics faster and more accurate, enabling reduction of unnecessary surgeries and improving the quality of patients' lives.
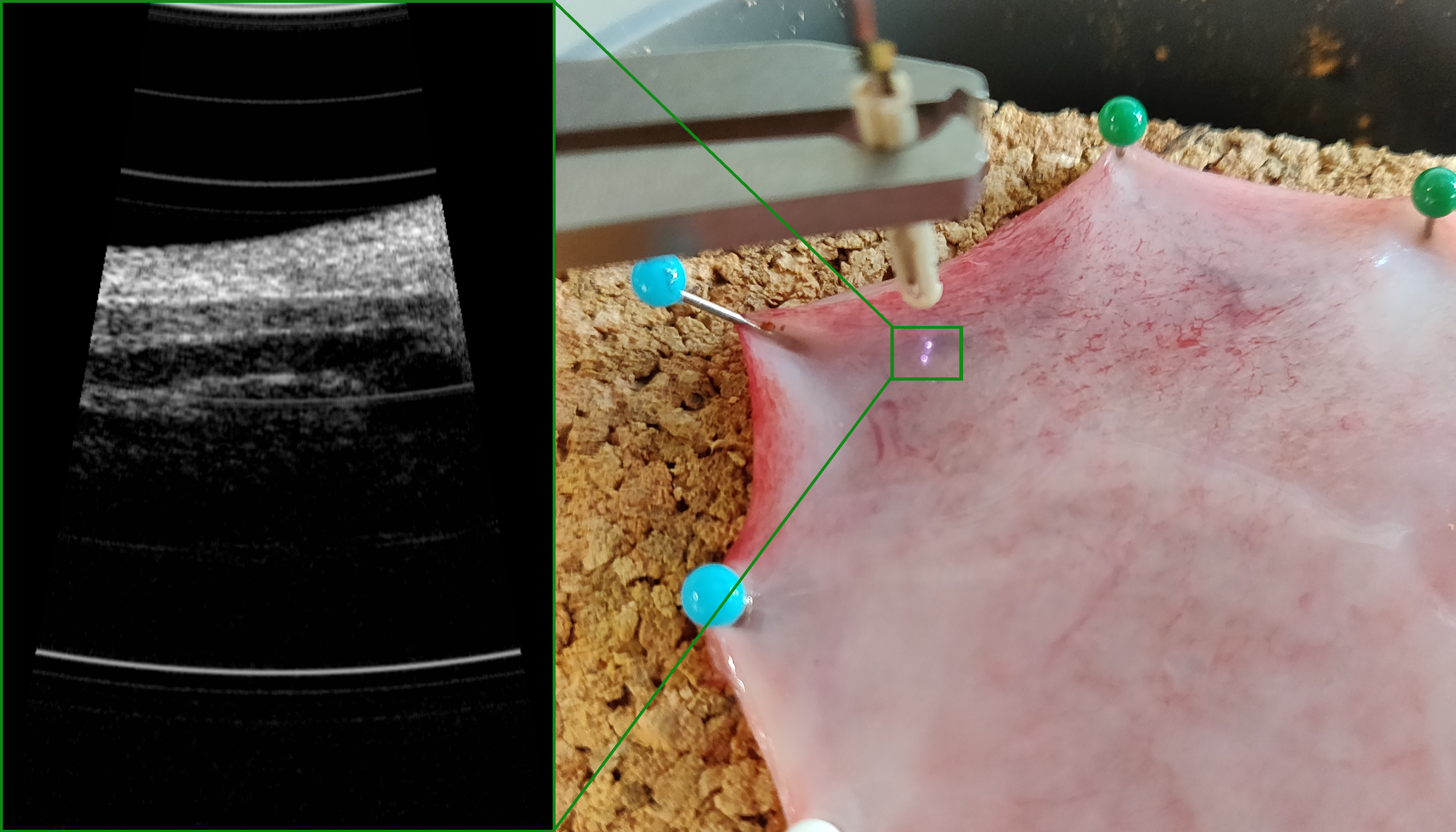
Scinvivo develops an imaging platform based on a forward looking OCT catheter to visualize the human morphological tissue structure a few mm’s deep with a high resolution. This approach fills the gap of the current image modalities and enables more precise minimal invasive surgeries and will significantly improve the diagnosis of cancer resulting in a precise personalized treatment.
Bladder cancer is chosen as the first field for the imaging platform. The urologists who reviewed the technology see this as a likely game-changer in their diagnostic routine with the potential to become the golden standard. Throughout Europe 10 Urologists, among others, Key Opinion Leaders in Urology, are all super enthusiastic and looking forward to working with this new technology after reviewing OCT images captured with the imaging platform.
Marijn van Os, CEO of Scinvivo: “With this investment, we will make the first steps in the clinics and show the incredibly high resolution tissue images captured with our platform.”
Maaike de Jong, CTO of Scinvivo: “ This investment enables us to perform first in vivo trials, to finalize our CE certification. We can now make a big step forward towards our goal of improving cancer diagnostics.”
Michel Simons, investor, and fund manager NSESF: ”Scinvivo is a diverse and experienced team, working on pioneering photonics technology, based on a sound IP position. The first market to address is bladder cancer. Being able to look into the tissue of hollow organs at near microscopic resolution, is possibly the key for the necessary improvement of diagnoses. We are happy to help with our investment and network.”
Wouter Keij, fund manager Innovatiefonds Noord-Holland: “The innovation Scinvivo develops can be an important addition to the way urologists work and an improvement for the diagnosis of different forms of cancer
NSESF
NSESF is a fund runned by Vectrix Participaties and fueled by its founders and a group of Dutch business angels on a deal-by-deal basis. The fund focuses on (pre) seed investments in high-tech ICT and Tech startups. https://www.vectrix.nl/venture-capital/vectrix-participaties/
Innovatiefonds Noord-Holland
The Noord-Holland Innovation Fund is an initiative of the Dutch province of Noord-Holland, University van Amsterdam, Hogeschool van Amsterdam, Amsterdam UMC, and Sanquin, with support from the European Union through the European Regional Development Fund. The Innovation Fund supports entrepreneurs in the Dutch province of Noord-Holland by financing innovation in the Proof-of-Concept phase by providing convertible loans. The first term of these loans is 2018-2023.
Scinvivo
Scinvivo is a MedTech startup, based in Eindhoven. www.scinvivo.com
Contact
Marijn van Os, CEO of Scinvivo, tel: +31 6 4609 3997
Maaike de Jong, CTO of Scinvivo, tel: +31 6 3605 4746
Press release distributed by Pressat on behalf of Scinvivo BV, on Thursday 10 March, 2022. For more information subscribe and follow https://pressat.co.uk/
Bladder Cancer Med-Tech OCT Innovation Investment Diagnostics Quality Of Life Deep-Tech Photonics Business & Finance Health Medical & Pharmaceutical
You just read:
Scinvivo secures 1 Million investment for in human clinical trials & CE
News from this source: